

It’s probably a good idea before we go further to reiterate exactly why we focus on vitamin K2 in this book. Why K2 and not K1? And is this case of mistaken identity really such a big deal?
Vitamin K2 has a wide range of biological roles, and has better overall health benefits. Vitamin K1 is primarily used for blood coagulation. Vitamin K2 can also be used in blood coagulation, but has many more functions.
Vitamin K2 can prevent calcium from being deposited into soft tissues like the kidney, where it can cause kidney stones, or into arteries where it can increase the risk of heart disease.
At the same time, vitamin K2 can help transport that same calcium into bones and teeth, improving bone density and strong teeth. Several observational and controlled studies have linked low levels of vitamin K2, especially of MK-7, with a higher incidence of fractures.
K1 does not have the same cardiovascular or bone benefits that K2 has. These functional differences between K1 and K2 have been demonstrated in recent studies.
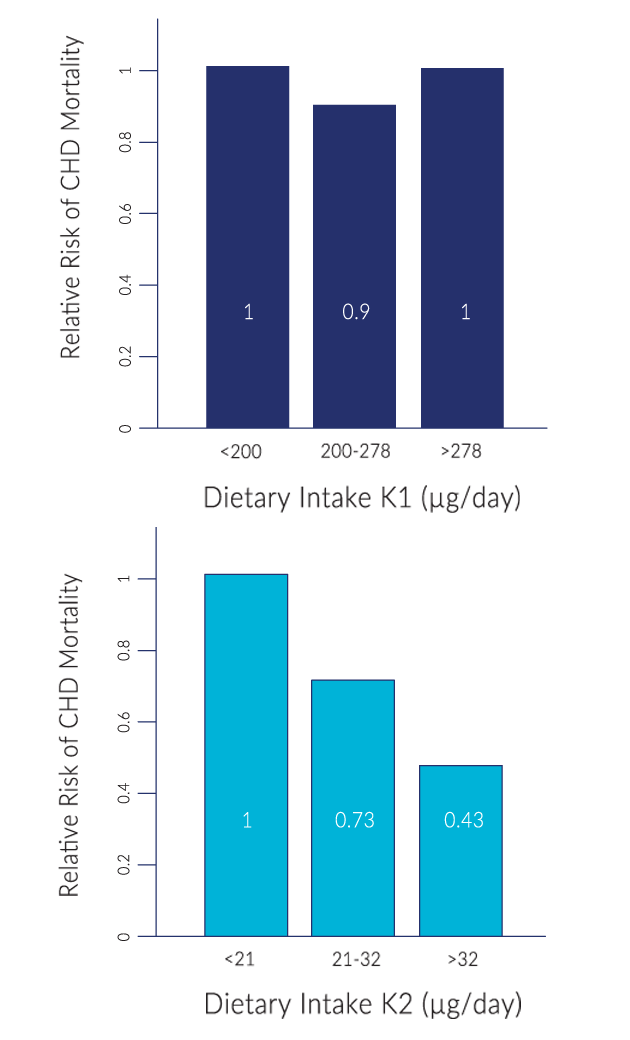
For example, in the large scale Rotterdam Study, where 4807 subjects were observed over 7-10 years, those in the highest vitamin K2 intake had a 57% reduction in the risk of dying from cardiovascular disease, compared to those with the lowest intake. And their risk of having severe aortic calcification was slashed by 52%.
Despite study participants consuming ten times more dietary K1 than K2, K1 intake showed no influence on aortic calcification or the risk of cardiovascular disease.
The main body of research has focused on vitamin K2’s influence on bone and heart health, but there are more studies underway to link K2 to other roles like insulin metabolism, increasing testosterone in males and facilitating growth in adolescence.
Overall, K2 is much better for general health than K1.
Because of its role in photosynthesis, vitamin K1 is found mainly in the leaves of plants. Its scientific name, phylloquinone, reflects that, phyllo meaning leaves in Greek.
 Present in leafy greens such as broccoli, kale and spinach, phylloquinone can be obtained through vegetables and vegetable oils. Vitamin K1 represents 90% of our total dietary intake in K vitamins.
Present in leafy greens such as broccoli, kale and spinach, phylloquinone can be obtained through vegetables and vegetable oils. Vitamin K1 represents 90% of our total dietary intake in K vitamins.
In contrast, menaquinones, or vitamin K2, are mostly microbial in origin. Simpler microbial organisms like bacteria produce and use vitamin K2 for many different metabolic processes. We can expect to find K2 in sources with a lot of microbial activity, like animal products such as fermented cheese and yogurt. (The first K2 isolates were in fact derived from putrefied fish meal.)
MK-4 form of vitamin K2 can be obtained from meat, dairy and eggs. MK-7 is mainly found in fermented foods like cheese, but it is particularly abundant in a Japanese dish of fermented soybeans called natto.
Unfortunately, the dietary intake of vitamin K2 has diminished over the past hundred years. Thanks to refrigeration and canning, we are eating less vitamin K2-rich fermented foods like cured fish and aged cheese. And thanks to industrial farming, animal sources aren’t great sources of vitamin K2 either. Animals have the ability to convert K1 to K2 from the grasses they graze on, but grain-fed animals are not consuming enough K1.
Unlike vitamin K1, it is possible to have a vitamin K2 deficiency without signs of overt bleeding or bruising. As vitamin K2 is heavily involved in cardiovascular and bone health, which are considered silent diseases, the signs of vitamin K2 deficiency often remain undetected until it is too late. Waning bone density or plaque build up in arteries may take years before they are detected.
It’s important to understand these differences because sometimes the differences aren’t so clear cut. And the occasional similarities tend to confuse the issue about which forms you need.
For example, there are studies that show that all vitamin Ks can promote healthy bones. However, vitamin K1 and MK-4 vitamin K2 show effectiveness only at high doses compared to MK-7 vitamin K2.
There’s also the argument that since animals break down and convert vitamin K1 into MK-4 vitamin K2, humans can too. Therefore, the kind of vitamin K we consume doesn’t matter. There are two problems with this argument. First, we don’t know enough about this conversion process to gauge how good humans are at turning K1 into K2 compared to other animals.
Secondly, if we could efficiently convert between the different forms, it shouldn’t matter which vitamin K to consume. Studies are showing otherwise. If there are no limitations, then research like the Rotterdam study would show that both K1 and K2 consumptions would have an effect on aortic calcification. However, only K2 demonstrated an effect.
So yes, vitamin K2 is quite different from K1. And it is something a lot of us could use more of.
Let’s see why.
Sometimes micronutrient deficiencies may not exhibit the usual array of signs and symptoms. Why does this happen?
Dr. Bruce Ames is a professor emeritus of molecular biology at the University of California Berkeley and a senior scientist at the Children’s Hospital Oakland Research Institute. In his research on degenerative diseases, he noted that deficiencies in various micronutrients may lead to DNA damage and cellular aging. He theorized that this was the consequence of something called triage allocation.
Organisms (like humans) commonly live through episodes of micronutrient shortages. Triage allocation theory hypothesizes that during those shortages, scarcer micronutrients are allocated towards urgent short-term survival at the expense of sustaining long-term health. For example, a shortage of iron will draw from stores in the liver before it draws critical iron stores in the heart.
In the case of vitamin K deficiencies, the body would allocate what little it has to support critical blood coagulation enzymes in the liver at the expense of bone building functions.
Triage theory: Rebalancing micro-nutrients for short-term survival at the expense of long term health.
Activator X’s secret identity is vitamin K2? And it plays a key role in bone and teeth health?
The notion of vitamin K2’s expanded role into bone health (and later cardiovascular health) seemed bizarre. How were these connected to vitamin K2’s original role as a blood coagulation aid? And what about all of these other health benefits we are just discovering now?
To explain that, we need to understand a little bit about how vitamins work on a fundamental level.
We are kept alive by trillions of chemical reactions that occur in the body. Carbohydrates are broken up and harvested for energy. New tissue is created. Cellular waste products are removed. New strands of DNA are made. This collection of chemical processes is called metabolism.
The speed that a reaction occurs will depend on factors like temperature, pressure, solubility and concentration of molecules. We use these factors every day. You might notice that sugar dissolves in hot water faster, or putting food in the refrigerator will slow the rate of decay. When you make a campfire, a hotter flame will use up wood faster.
Our metabolism needs to occur at a certain speed to stay alive. But we don’t have the liberty of turning the body into a raging furnace to speed up these reactions (not without damaging the body). That’s where enzymes come in.
Enzymes are bits of protein that catalyze and regulate almost all metabolic reactions. As catalysts, they reduce the energy needed to spark a chemical reaction. They speed up the rates of reaction. Without enzymes, reactions that would normally take milliseconds might take hours or days.
Some enzymes require helpers called cofactors to ignite a reaction. Without a cofactor bound to its structure, an enzyme may float dormant and inactive, unable to catalyze any reactions. All vitamins serve as cofactors for enzyme proteins. When a vitamin is absent, the proteins that depend on it are left inactive.
 How long would reactions take if they proceeded spontaneously without the presence of enzymes? Dr. Richard Wolfenden, an alumni professor of biochemistry & biophysics at the University of North Carolina, posed this question. In a 1995 study, Wolfenden reported that without enzymes, the process of synthesizing DNA and RNA would take 78 million years. A subsequent study in 2008 found that producing cellular hemoglobin in the absence of enzymes would be thirty times slower, with a half-life of 2.3 billion years. That’s half the age of Earth!
How long would reactions take if they proceeded spontaneously without the presence of enzymes? Dr. Richard Wolfenden, an alumni professor of biochemistry & biophysics at the University of North Carolina, posed this question. In a 1995 study, Wolfenden reported that without enzymes, the process of synthesizing DNA and RNA would take 78 million years. A subsequent study in 2008 found that producing cellular hemoglobin in the absence of enzymes would be thirty times slower, with a half-life of 2.3 billion years. That’s half the age of Earth!
All vitamins activate protein enzymes a little differently.
K vitamins activate proteins through a process called gamma-glutamyl carboxylation.
Sounds complicated? Don’t worry – it’s just a technical way of saying that vitamin K attaches carbon dioxide to a specific site on a vitamin K dependent protein.
Let’s break it down.
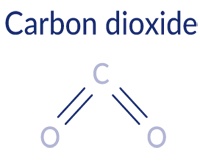 We are familiar with CO2 as carbon dioxide, the stuff we exhale. Inside the body, CO2 is dissolved in our blood and tissues, making it an ion with a negative charge. We call this form a carboxyl group (COO-).
We are familiar with CO2 as carbon dioxide, the stuff we exhale. Inside the body, CO2 is dissolved in our blood and tissues, making it an ion with a negative charge. We call this form a carboxyl group (COO-).
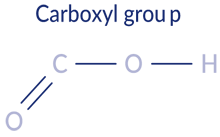 Carboxylation is the act of attaching a carboxyl group to another molecule. Decarboxylation is the process of removing a carboxyl group from a molecule.
Carboxylation is the act of attaching a carboxyl group to another molecule. Decarboxylation is the process of removing a carboxyl group from a molecule.
Proteins are made up of chains of amino acids. Glutamic acid is one such amino acid, made up of three carbon atoms and one carboxyl group. This is the part of the protein that vitamin K attaches a carboxyl group to.
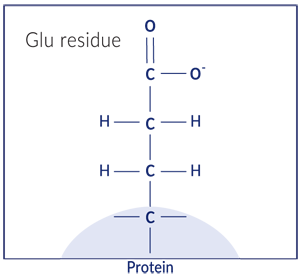
*When glutamic acid is part of a protein, it’s also known as a glutamate or “Glu” residue.
Gamma just refers to which carbon atom in the glutamate residue we are attaching the carboxylic acid to. Sort of like a street address. In this case, gamma refers to the third carbon in the side chain.
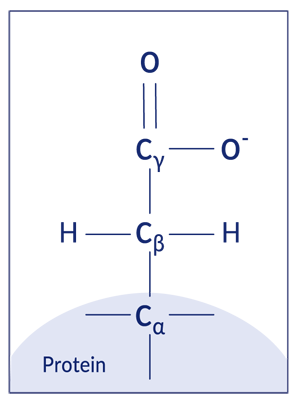
Let’s put it all together. Vitamin K attaches a carboxyl group to the third carbon of a glutamate residue. This modified glutamate structure is called a gamma-carboxyglutamate or “Gla” residue.

Once activated, this Gla residue has two carboxyl groups attached to the gamma carbon. This gives the residue two negative charges, which is ideal for binding (or attracting) calcium ions, which have two positive charges. This calcium manipulation, as we’ll see later, is the key.
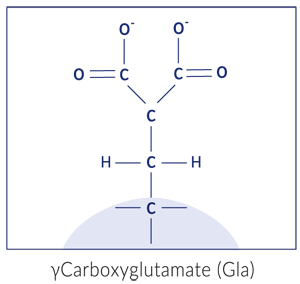
When you hear biochemists talk about vitamin K dependent proteins, you may hear them refer to the protein as gamma-carboxylated (gla/gc) or undercarboxylated (uc) . They’re just indicating whether the enzyme is activated or not.
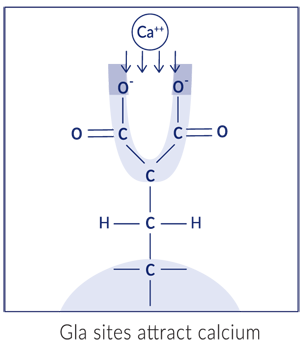
Vitamin K dependent proteins (VKDPs) are found throughout the body in different tissues. Scientists have now identified 17 different VKDPs that undergo the same carboxylation process but fulfill different functions. Some help with building up the bone matrix. Some prevent arteries from calcifying. Others increase insulin sensitivity, renew tissue, control cell growth or sweep dead cells away. They are present in a wide variety of tissues, such as bone, kidney, placenta, pancreas, spleen, lung, and blood vessel walls.
We haven’t even figured out what some VKDPs do yet, and we are still discovering new VKDPs.
Seven VKDPs, known as clotting factors, regulate blood coagulation. They are made in the liver and released into the bloodstream, inactive until needed. When injury occurs, these clotting factors activate, transforming fibrinogen (also present in plasma) into fibrin. When fibrin is combined with blood platelets, it forms a clot.
(Each VKDP in the clotting cascade ultimately leads to the transformation of prothrombin into thrombin. Thrombin is the active enzyme that turns fibrinogen into fibrin clots.)
While vitamin K2 is circulated to other tissues and organs in the body, vitamin K1 is found primarily inside the liver, which is why K1’s role is so tightly bound to blood coagulation.
This mechanism of action is well understood and acted upon by the medical community. Newborns are routinely given vitamin K1 injections to prevent a fatal bleeding disorder called ‘vitamin K deficiency bleeding’. Many coumarin-based anticoagulants like warfarin work by preventing vitamin K from being recycled in the body.
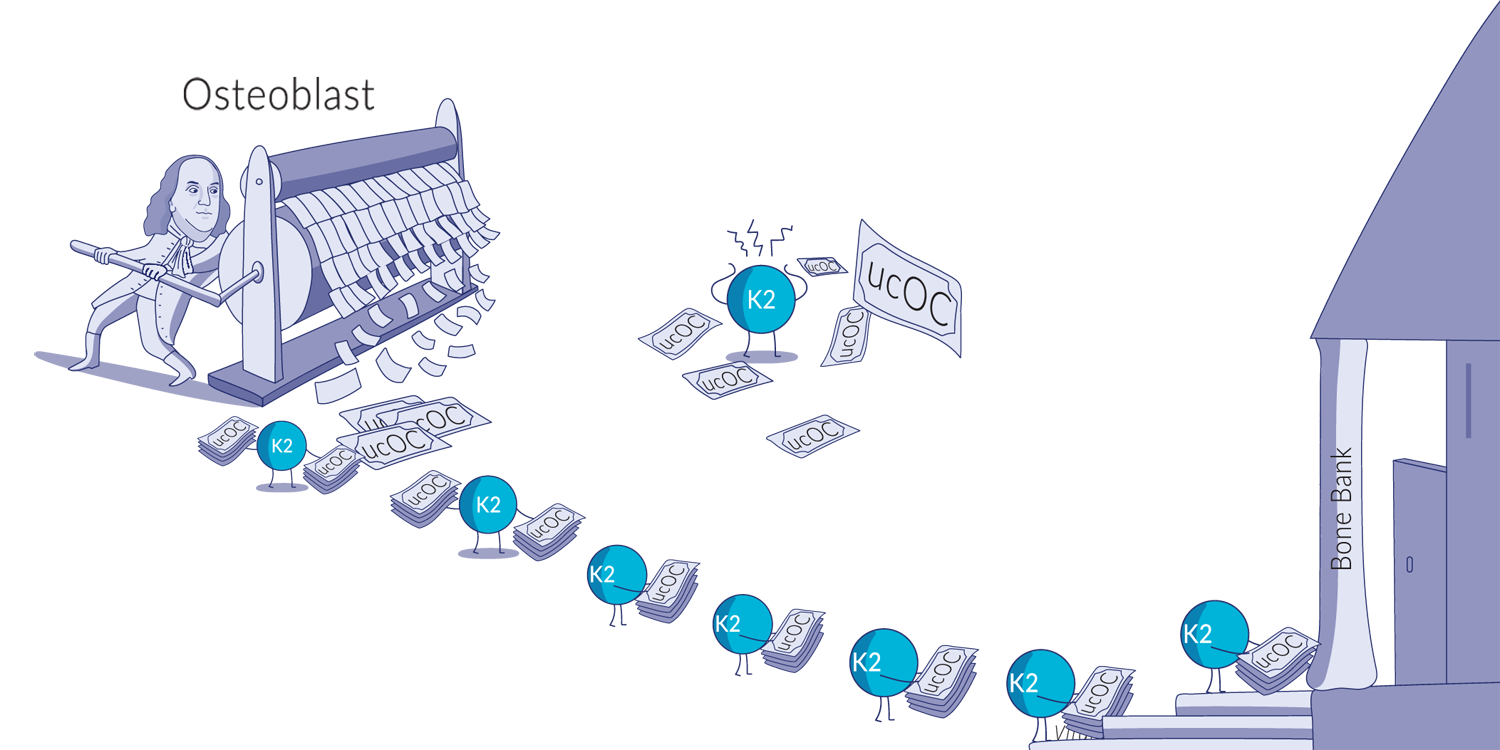
Vitamin K2 is used in the activation of a protein called osteocalcin. This VKDP, discovered in 1975, was the first hint of vitamin K2’s greater role in the body.

Osteocalcin is produced in bones and teeth where, when activated by vitamin K2, helps to transport and integrate calcium into the bone matrix. Carboxylated osteocalcin binds to the calcified matrix of bone tissue. Although one of the first functions identified, osteocalcin’s roles in bone mineralization and architecture are still being investigated.
One recent line of investigation has suggested that osteocalcin, along with another protein called osteopontin, helps to prevent crack growth in bone fractures. Carboxylated osteocalcin and osteopontin combine to form elastic collagen fibrils in bone. These fibril bands act as shock absorbers or springs, making bones a little more flexible.
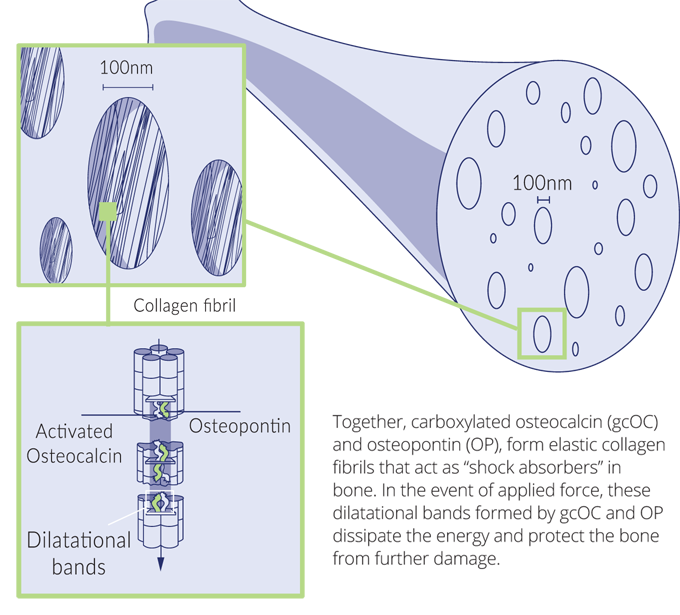
When a bone is subjected to bending force, tiny 100nm voids appear in the bone matrix. The osteocalcin/osteopontin fibrils stretch to form dilatational bands in the voids, dissipating the energy and preventing greater damage. When the stress subsides, the bands can close and the nanoscale holes can be repaired.
Carboxylated Osteocalcin
Undercarboxylated Osteocalcin
While a carboxylated osteocalcin deficiency may not impact bone mineral density, it might affect the bone’s ability to withstand force and pressure, increasing the risk of fractures. It’s like comparing two concrete bridges: one built with reinforced concrete versus one with no steel bars. While they may look the same and have the same mass, the one built with reinforced concrete will be able to withstand more weight-bearing loads.
 Osteocalcin-lacking mice suffer other health problems. They are overweight and hyperglycemic, due to their poor glucose tolerance, insulin levels and insulin sensitivity. Males have poor fertility, shown by lower testosterone levels and lighter weighing reproductive organs. Behavioural testing showed increased behaviours linked to anxiety and depression. Many of these symptoms have been observed in cross-sectional studies in humans, with significantly reduced blood osteocalcin levels reported in populations like pre-diabetics, untreated diabetics and obese children.
Osteocalcin-lacking mice suffer other health problems. They are overweight and hyperglycemic, due to their poor glucose tolerance, insulin levels and insulin sensitivity. Males have poor fertility, shown by lower testosterone levels and lighter weighing reproductive organs. Behavioural testing showed increased behaviours linked to anxiety and depression. Many of these symptoms have been observed in cross-sectional studies in humans, with significantly reduced blood osteocalcin levels reported in populations like pre-diabetics, untreated diabetics and obese children.
Our bones produce, store, and supply the osteocalcin needed for these hormonal and metabolic functions. When needed, bone cells called osteoclasts decarboxylate and release stored osteocalcin from the bone into the blood. From there, this undercarboxylated-osteocalcin can travel to and influence different tissues in the body.
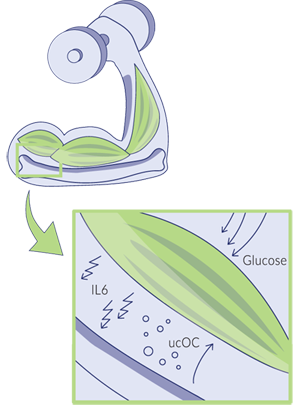 When we exercise, our muscles secrete an inflammatory hormone called interleukin-6 (IL-6), which stimulates the decarboxylation and release of osteocalcin from our bones. This osteocalcin circulates to the skeletal muscles and increases the uptake of glucose and fatty acids, improving energy utilization, insulin sensitivity and muscle strength.
When we exercise, our muscles secrete an inflammatory hormone called interleukin-6 (IL-6), which stimulates the decarboxylation and release of osteocalcin from our bones. This osteocalcin circulates to the skeletal muscles and increases the uptake of glucose and fatty acids, improving energy utilization, insulin sensitivity and muscle strength.
Another well-studied VKDP is matrix Gla protein (MGP). MGP is found throughout the body in vascular smooth muscle cells, soft tissues, and bone cartilage.

If you recall from the previous section, when VKDPs are carboxylated by vitamin K2, they are able to bind to calcium. Matrix Gla protein uses this property to transport and deliver calcium to where it is needed, like the bone matrix and teeth where roughly 99% of all body calcium is stored.
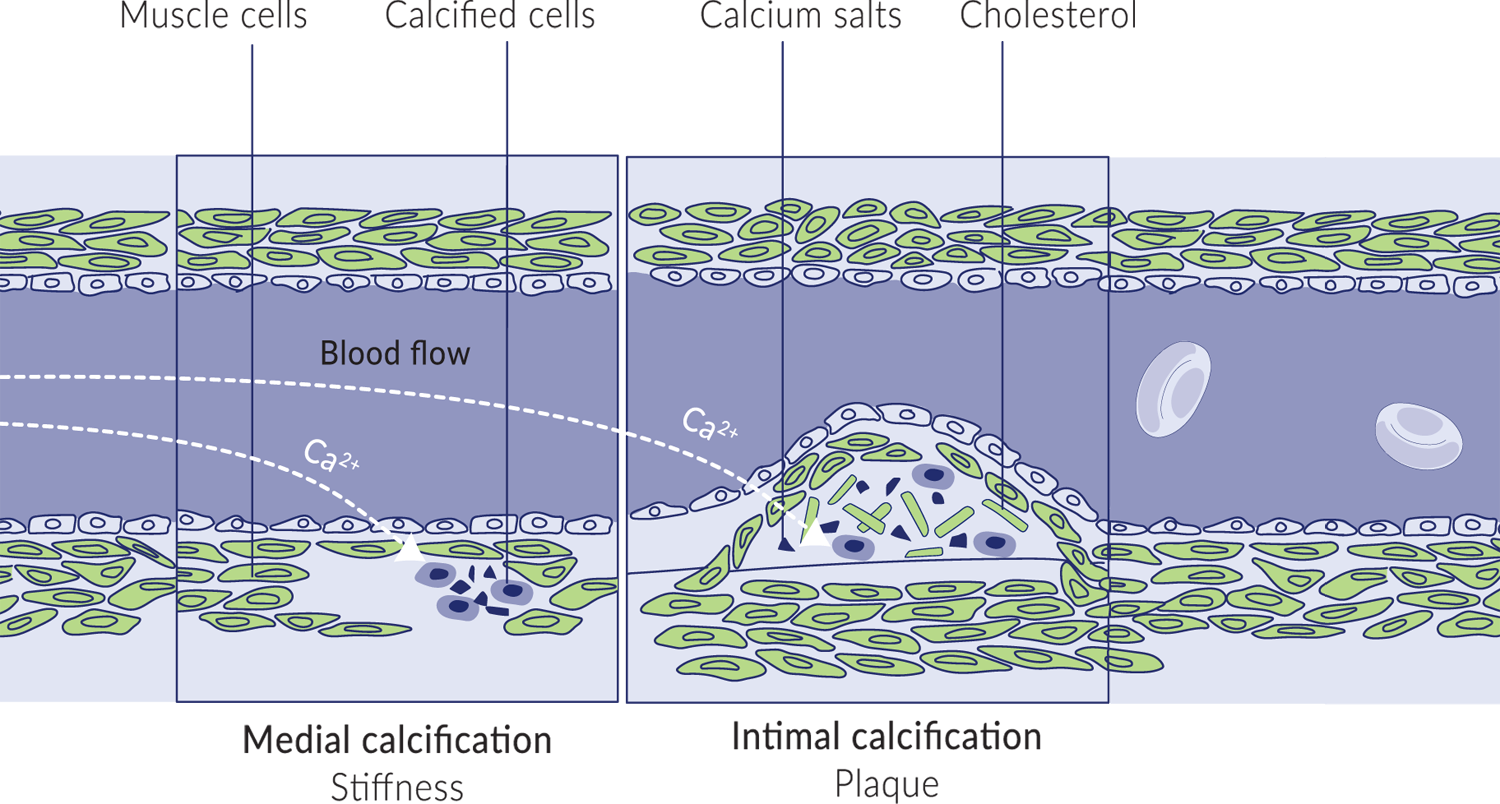
This binding also prevents unwanted calcification – calcium build ups in soft body tissue, blood vessels, and organs. This calcification can disrupt our biology and lead to very dangerous complications such as heart disease and chronic kidney disease.
Calcium ions are very reactive. When dissolved in blood, calcium ions can react with phosphates to form calcium salts. With sufficient numbers and size, calcium salts can harmfully accumulate on soft tissues. For example, calcium salts can accumulate in the kidneys, forming kidney stones.
Calcium salts can also accumulate inside blood vessels. They can accumulate in and harden the elastic muscle cells of arteries. They can also build deposits along the inner walls, leading to atherosclerotic plaques. Both kinds of calcium build ups can severely obstruct blood flow. Indeed, coronary artery calcification is an independent predictor of cardiovascular disease.
Calcification is not limited to the kidney and blood vessels. Since calcium is found in every cell, these accumulations could occur almost anywhere in the body, including joints, tendons, muscles, organs and even the brain. This is why many organs in the body produce matrix Gla proteins…and vitamin K2 to activate them.
Vitamin K2 carboxylated MGP prevents these salts from forming by binding calcium ions in the bloodstream. Calcium bound to MGP is unable to interact and form salts with phosphorus, preventing mineralization.
The relationship between vitamin K2 and vascular calcification has been shown in several studies. The previously mentioned Rotterdam study showed that those who had the highest intake of vitamin K2 supplementation over a span of 7-10 years were 52% less likely to suffer arterial calcification. Another cohort study with 16,057 women suggested that for every 10mcg of K2 consumed per day, the risk of coronary heart disease was reduced by 9.1%. In healthy, postmenopausal women, there was an association between undercarboxylated-MGP and coronary artery calcification. Similar observations were made between vitamin K deficiencies and patients with chronic kidney disease. Vitamin K2 supplementation in hemodialysis patients decreased inactive MGP in the blood.
The formation of calcium salts can also impede the mineralization of bones and teeth. Calcium phosphate salts that are too large are unable to pass between proteins that make up the bone matrix. Activated MGP, by preventing the formation of calcium salts, allows calcium to penetrate the bone matrix and properly mineralize the bone.
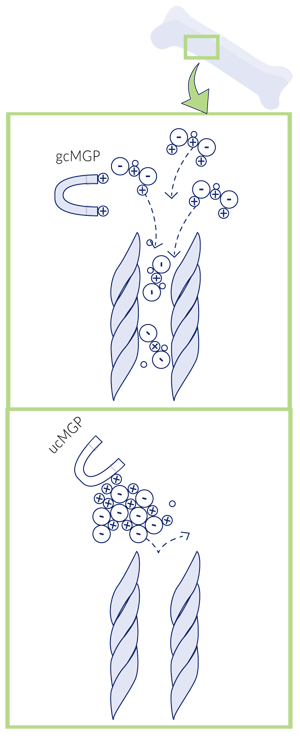
Carboxylated MGP keeps calcium salts small, so they can pass through the bone matrix.
Many people know that vitamin D3 is essential for building and maintaining a healthy skeleton. Vitamin D3, after all, is essential for absorbing dietary calcium in the intestinal tract. Vitamin K2, by activating MGP, ensures that calcium traveling in the bloodstream reaches the bone and is properly incorporated into the bone matrix.
Childhood and adolescence are both periods of intense skeletal bone growth. For bones to lengthen and grow, cartilage at the growth plates of bones produces new cartilage that then calcifies and becomes bone. Once growth is complete, the cartilage in the growth plate calcifies.
This suggests that vitamin K2 is crucial for growing children and adolescents.
Growth plate cartilage cells produce MGP to protect itself from calcification. However, if there isn’t enough vitamin K2 activated MGP, there is a risk of premature calcification, stopping growth too early.
This suggests that vitamin K2 is crucial for growing children and adolescents.
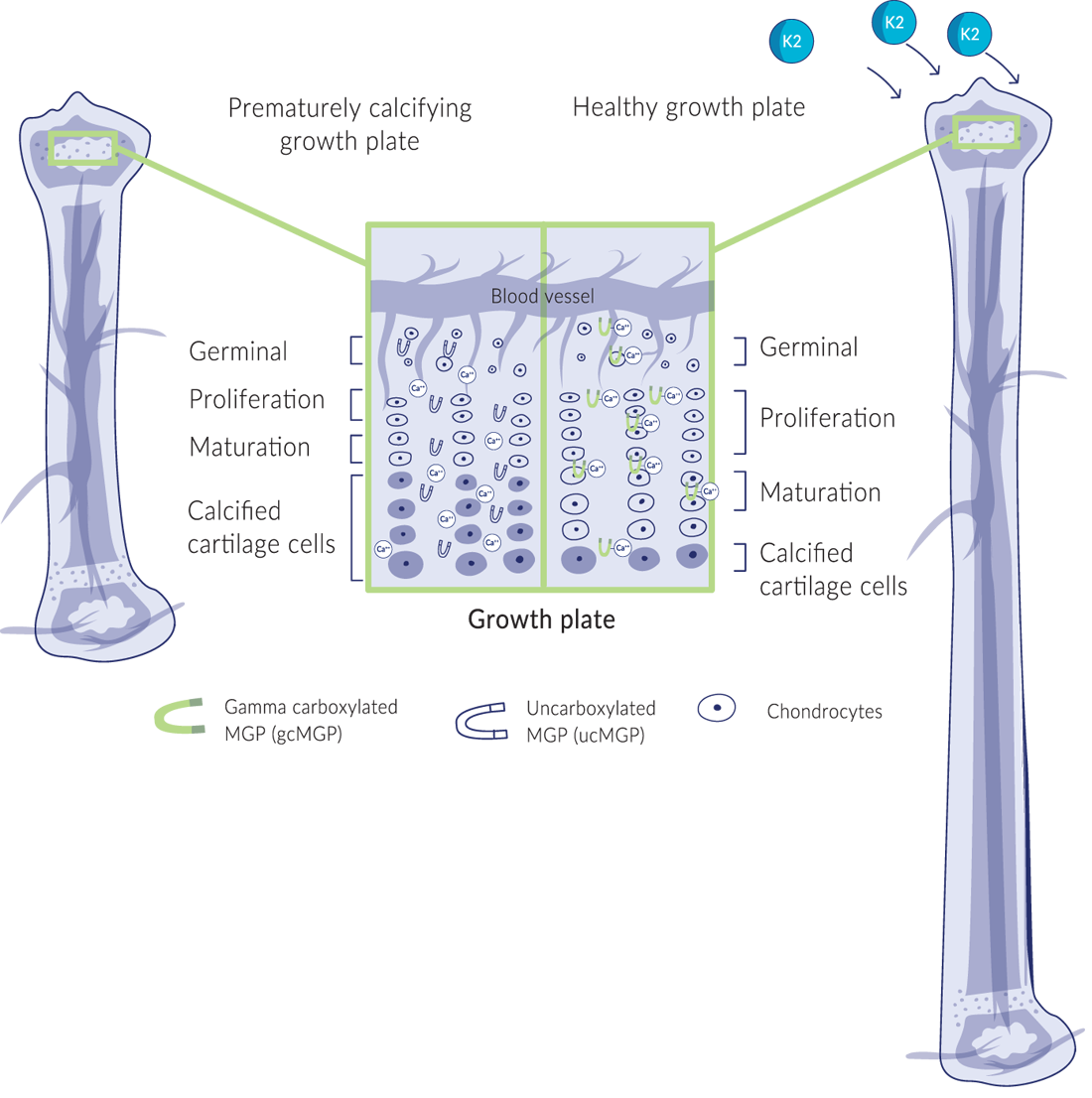
Growth plate cartilage cells (known as chondrocytes) produce MGP to protect themselves from early calcification. The growth plate is divided into several distinct regions. Near the head of the bone, the cartilage cells are germinating and proliferating, followed by a zone where they mature and grow in cell size. Towards the bottom of the growth plate, cartilage cells absorb calcium and form the hardened bone structure. When there isn’t enough vitamin K2-activated MGP circulating, too many cartilage cells may prematurely calcify, stopping bone growth too early.
Besides the above, there are several more vitamin K-dependent proteins that still need to be more fully studied. They include Gla-rich protein (GRP) which might act as an anti-inflammatory and also inhibit calcification, periostin which may be involved in tissue regeneration and healing, and growth arrest-specific protein 6 (Gas6) thought to be involved with cell proliferation.
Several studies have demonstrated that vitamin K2 supplementation increases the activation of osteocalcin and MGP. In a study of 244 healthy postmenopausal women, taking 180mcg of MK-7 vitamin K2 daily for three years limited the age-related declines in bone mineral density and bone strength in the lumbar spine and femoral neck. Reduced loss of vertebral height in the lower chest region was also observed. Results also showed a significant decrease in arterial stiffness.
Necessary cookies are absolutely essential for the website to function properly. This category only includes cookies that ensures basic functionalities and security features of the website. These cookies do not store any personal information.
Any cookies that may not be particularly necessary for the website to function and is used specifically to collect user personal data via analytics, ads, other embedded contents are termed as non-necessary cookies. It is mandatory to procure user consent prior to running these cookies on your website.
Analytics cookies help us understand how our visitors interact with the website. It helps us understand the number of visitors, where the visitors are coming from, and the pages they navigate. The cookies collect this data and are reported anonymously.
These cookies collect information about how visitors use a website, for instance, which pages visitors go to most often, and if they get error messages from web pages. These cookies don’t collect information that identifies a visitor.
The “Other” category cookies help us provide our visitors proper functionality of the website such as online quizzes or embedded videos.
Advertisement cookies help us provide our visitors with relevant ads and marketing campaigns.
Learn about how vitamin K2 works, why it’s important, and how it can help you.
